Subject Code & Title :- BIOS5004 Microbiology
Assessment Type :- Assignment
Scenario :- You are employed by a pharmaceutical company, where all research in your Department has recently been redirected to explore new antiviral candidates for the inhibition of SARS-CoV-2 corona virus and hence the treatment of Covid-19 patients. As one of the virologists you have been searching for promising compounds.
BIOS5004 Microbiology Assignment

Background :-
Several drug candidates have been explored over the past 2 years for treatment of severe Covid-19 hospitalise cases including two that have shown particular promise:
Favipiravir is a nucleoside analogue that can act as a mimic of both guanosine and adenosine. It has been documented as having inhibitory effects against a range of viruses that have an RNA genome. It is not currently licenced for use against Covid-19.
Molnupiravir is a synthetic nucleoside derivative that gained approval for medical use in the UK in November 2021 and in the US a month later. However the approval is only for emergency use in certain populations because the drug’s efficacy is limited and there are concerns that the mechanism of action of molnupiravir could in advertently pro long the COVID-19 pandemic.
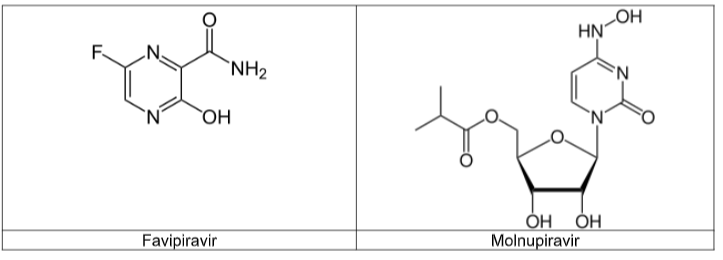
Because of the limitations to the use of molnupiravir, your company is keen to find alter natives that can be used safely and effectively in all cases of severe Covid-19 disease.
However your company will only receive approval to pursue clinical trials with compounds that show statistically significant superior antiviral effects compared to molnupiravir.
Preliminary screening by your company of over 10,000 compounds in their archives has high lighted three drug compounds that have demonstrated inhibition of SARS-COV-2 in basic in vitro tests.
You now intend to test these three compounds more fully, in a cell-based assay that will allow you to consider the level of virus inhibition in direct comparison with molnupiravir.
Experimental work :
You are using a cell line known as Vero/hSLAM (Ono et al., 2001 1 ) which you have sourced from the ECACC General Cell Collection (part of Public Health England), and which is fully permissive for SARS-CoV-2 replication.
You routinely culture these cells as monolayers, which means that they adhere as a single layer of cells on the inside wall of a specialised tissue culture (plastic) flask.
The cells grow under a thin depth of nutrient liquid known as a culture medium.
Your use a medium called Earle & #39;s Minimum Essential Medium (EMEM) which must be supplemented with Fetal Bovine Serum (FBS) to a final concentration of 7% FBS to main tain healthy cells and to promote mitosis.
BIOS5004 Microbiology Assignment
The cells grow best at 37°C and in an incubator with a 5% CO 2 atmosphere to maintain the pH of the cells and medium as they metabolise nutrients and release by products.
Below is an outline of the experiment – read it carefully and then answer the questions at the end.
Submit your work as a single document into the Turnitin link on the module Moodle page.
Day 1 – routine cell culturing
You have been maintaining monolayer cultures of Vero/hSLAM cells in small plastic cell culture flasks. You begin your experiment by establishing enough of these flasks to have plenty of cells to work with. Every thing that you do is performed in a sterile laminar flow hood and you practice high-level aseptic technique.
Taking cells from a flask that has been set up a few days ago you replace the culture medium in the flask with 3 mL of phosphate-buffered saline (PBS).
You scrape / dislodge the cells from the flask wall into this solution PBS is an isotonic solution that preserves the cells for a short period of time.
You count a sample of the cells suspended in PBS using a haemocytometer to determine the cell density of this temporary cell suspension.
Using this cell density you calculate the volume of cell suspension that you would need to add to a new flask to establish a starting culture containing 4 x 10 5 cells per flask.
You add 20 mL of FBS-supplemented EMEM mixing these components together before you add them.
You set up 3 of these flasks and incubate the 3 new cultures at 37°C in an incubator with a 5% CO 2 atmosphere, checking them daily for cell growth.

Day 4 – establishing the test cultures
The cells reach over 95% confluence by Day 4 meaning they are ready to use. You use one of the flasks to repeat the procedure above in order to keep the cell line going (i.e. you set up 3 new flasks just as you did on Day 1).
You aim to set up a series of test culture plates that allow you to make small replicate cultures in a series of wells.
o Each test culture plate has 24 wells for cell culture
o You want to set up 4 of these 24-well culture plates
o Every culture well needs to have 2 x 10 4 cells added
o Every culture well needs 1 mL volume in total.
You intend to make a single master mix cell suspension to do this to promote accuracy consistency and to minimise pipetting errors.
This master mix must contain the right number of cells to allow you to set up all your test cultures in one go
the right volumes of EMEM and FBS to allow you to set up all your test cultures in one go
In other words you need to make just one single mixture of cells EMEM and FBS from which you simply pipette 1 mL into every well of the four 24-well plates.
Once the test plates are set up, you incubate the test culture plates at 37°C and in an incubator with a 5% CO 2 atmosphere for 2hr to allow the cells to adhere to the bottom of the wells.
Day 5 – Infecting cultures with virus and testing candidate antivirals
After you incubate the test cultures for 2hr you infect every well (i.e. every monolayer culture with SARS-CoV-2 at an MOI of 2.0 pfu / cell. Once the cells are infected you incubate them in the presence of different drugs.
The right volume of virus calculated to achieve the required MOI is added to every well of all the cell culture test plates.
The plates are incubated at 37°C for 2hr to allow the virus to attach to and penetrate the cells.
The inoculum the medium containing the virus) is removed from every well and the mono layers are quickly washed with medium containing no virus.
1 mL of fresh FBS supplemented EMEM media is added to every well either molnupiravir or one the 3 candidate compounds you have found promising in earlier tests.
Each compound is tested across a 24 well plate at 3 concentrations meaning that each concentration is tested in 6 wells.
Each plate contains a negative control which comprises 6 wells incubated with FBS supplemented EMEM containing no drug.
The layout for each test plate is as follows. Remember every well contains a known number of cells and a known amount of virus. All that differs between the wells is the concentration of the drug. Each test plate there are 4 is set up with one of the drugs.

Day 8 – harvesting virus and the plaque assay
The cells are incubated for 3 days at 37°C and in an incubator with a 5% CO 2 atmosphere to main tain the pH of the cells and medium.
The liquid medium of each well is harvested and stored at 4°C. Over the coming weeks you will test every harvested sample using a plaque assay, to determine the amount of virus that has been released from the infected cell cultures. You hope that one of your test drug candidates will have had a significant effect on the amount of virus released.
Remember:
there were 4 drugs tested molnupiravir and your 3 unknown drug candidates
each drug was tested at 3 different concentrations
each concentration was tested using 6 replicate wells.
That’s a lot of plaque assays! Each was done in the same way as follows:
A 24-well plate of test cultures was set up using the same cell numbers as for Day 4.
The harvested medium to be tested for virus released from infected cultures was diluted in 10-fold series up to 10 -9 . Dilutions were made in EMEM just before use.
Each virus dilution was tested in triplicate by infecting 3 wells 3 cultures with 0.1 mL of the diluted virus.
After 2hr this virus inoculum (0.1 mL) was removed and replaced with 1 mL of FBS- supplemented EMEM containing 1 % molten agarose which was allowed to set.
The culture plates were in cubated for 3 days at 37°C and in an incubator with a 5% CO 2 atmosphere.
BIOS5004 Microbiology Assignment
At the end of this period the cell monolayers were stained in order to highlight virus plaques and these were counted manually and recorded.
For each plaque assay the mean virus titre was determined using the 3 replicates and the results were recorded you will find the virus titres on the following page.
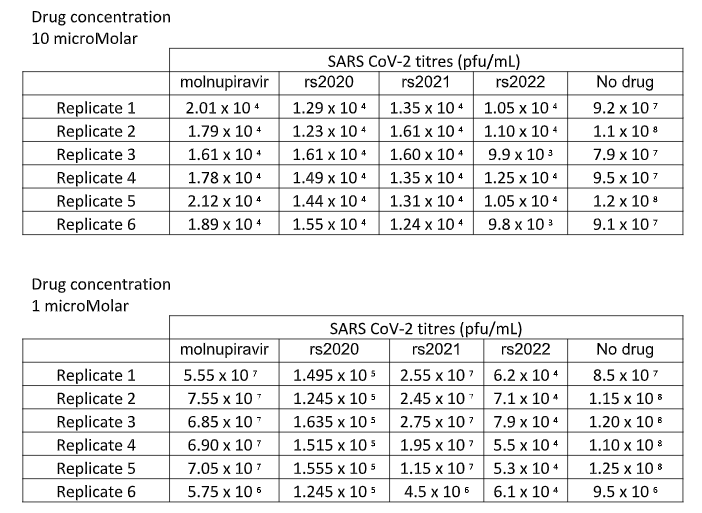
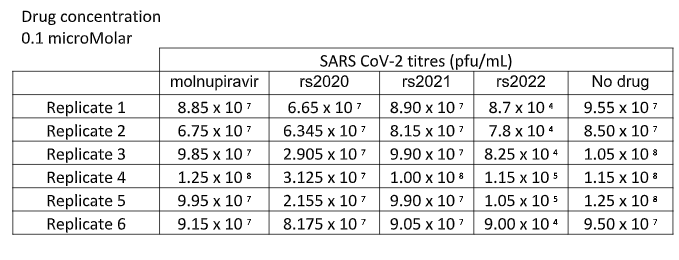
BIOS5004 Virology Resit 2022 questions
Answer questions (a – g) and present / evaluate the experimental data as per the instructions.
Background
a) Research and explain the mechanism of action of favipiravir (maximum 250 words).
Make sure you use your own words, to showcase your understanding. Cite & reference your sources accurately and appropriately.
b) Research and explain the mechanism of action of molnupiravir (maximum 250 words).
Make sure you use your own words, to showcase your understanding Cite & reference your sources accurately and appropriately.
Experimental work
For all of the calculations try to imagine that you will do this work in the lab yourself using familiar equipment With that in mind:
Show your complete workings to gain full marks for each calculation. Scan these into your submission document.
Apply a sensible number of decimal places to the final answer representing genuinely achievable volumes that can be manipulated with accuracy using equipment that you have used in your practical classes. For example it is not possible to measure 1.23456789 mL accurately.
You may find it useful to revisit the quizzes set up for you during the semester to help you with these calculations. All of the calculation types required here were covered in the quizzes and most of them are simply variations of the numerical assessment questions you’ve done previously in your 1 st year Biochemistry modules.
Day 1 – routine cell culturing
The cell density of the temporary cell suspension (in 3 mL PBS) is 2.6 x 10 6 cells / mL
c) What is the total number of cells in the PBS suspension?
d) You want to establish a new culture in a new flask with the cell density given in the protocol. What volume of the cell suspension is needed?
e) What volumes of EMEM and FBS must you mix together to create the required volume of FBS supplemented EMEM?
Day 4 – establishing the test cell cultures
The cell density of the temporary cell suspension is 2.73 x 10 6 cells / mL.
f) Calculate the volume of each component needed in the master mix to allow you to add the right number of cells, and have the right level of FBS in every one of the culture wells in other words to set up the right number of 24-well plates
i. cell suspension
ii. FBS
iii. EMEM
Day 5 – infecting the test cultures
The titre of your virus is 3.19 x 10 6 pfu / mL.
g) Calculate the volume of virus needed to infect every test well every test culture at the required MOI. Express your final answer in microlitres (mL).
Experimental outcomes – Plaque assay results
Introduce present describe and evaluate the experimental data offering a suitable evidence based conclusion.
Present a single figure with a single legend.
Ideally you should plot all the results on a single graph, such that it forms a single figure with a single legend. Alternatively you can separate graphs if you wish but this is not such a good approach to the presentation of this data.
NOTE 1 –
you must use an established computer package for plotting the graph(s) hand drawn graphs will not be accepted because you must plot a log-scale graph and add accurate error bars representing the standard deviations for each mean value. You are expected to read the original practical booklet for guidance and you may find it useful to listen to the relevant recorded lectures.
NOTE 2 – if you choose to plot separate graphs think carefully how to cluster the data in each graph and arrange the multiple graphs into a single figure with a single legend. The axes of every graph must match each other in scale if you present them in this way – you will need to do this as you create the graphs.
Offer suitable narrative text
Give the reader some context about what the figure is representing and describe key points or themes that the data reveals. It’s not enough to simply offer a figure with out some
ac companying description and you will gain limited marks if you don’t give any sensible text.
Use a suitable statistical test to evaluate the results and identify if there are any significant
variations between the yields of virus for each of the drugs tested. You can use Excel or R to
perform the test if you wish.
BIOS5004 Microbiology Assignment
NOTE 3 – include the full statistical calculations at the end of the document.
Be careful here – your goal is to compare potential drug candidates with the current approved drug molnupiravir. You may also want to compare each new candidate to each other but you don’t want to compare any of them to the negative control no drug. Make sure your stats ONLY analyse data from replicate titre values derived from ;plaque assays performed with the drugs present (at the different doses).
Summarise the outcomes of your experimental and statistical results within the narrative text and discuss what they mean. Remember your aims here and explain if and why the results indicate if any of the potential drug candidates are worthy of further investigation.
ORDER This BIOS5004 Microbiology Assignment NOW And Get Instant Discount
Read More :
Unit 17 Microbiology And Microbiological Techniques Assignment – UK



